Understanding Zwitterionicity.
Nature’s way of avoiding interactions is to create zwitterionic surfaces. In fact, zwitterionicity is the basis for modern antibacterial surfaces. Designing drugs with zwitterionic properties where only the targeting ligands protrude from the drug core prevents non-specific, off-target binding and promotes fast elimination of unbound drug from the body through the kidneys and into urine.
Curadel’s scientists have been working for over twenty years to develop an entirely new class of zwitterionic drugs.
Whereas most drugs are designed to pass through cell membranes, zwitterionic drugs are the opposite. They are polyionic, with alternating fixed strong positive and negative charges, yet are electrically neutral with a net zero surface charge. By balancing this charge over entire molecular volume, zwitterionic drugs are not “sticky;” they do not bind non-specifically to cells or tissues. They are rapidly cleared by the kidney, being eliminated into urine. These findings were validated and published in a landmark paper in Nature Biotechnology in 2007 in collaboration with a future Nobel laureate.
Zwitterionicity creates a net-zero-charged forcefield around the drug core.
- Repels binding except for protruding ligands to protect healthy tissue
- Targets and rapidly kills tumor cells with purposefully selected isotope
- Rapidly cleared through the kidneys
Zwitterionicity transforms radiotherapy outcomes.
Current and emerging targeted alpha therapies (TATs) rapidly and effectively reduce tumors. Yet data show they do not currently deliver extended survival benefits for cancer patients. Why? They are not designed to prevent tumor resistance, which is the major contributor to treatment failure and resulting mortality.
Curadel addresses tumor resistance by combining the strong firepower of radiotherapy, unique binding, and proprietary zwitterionic chemistry.
Rapid, complete tumor eradication prevents resistance.
Monomer products target only one cell type, despite most tumors being heterogeneous, leading to resistance through clonal evolution and poor survival.
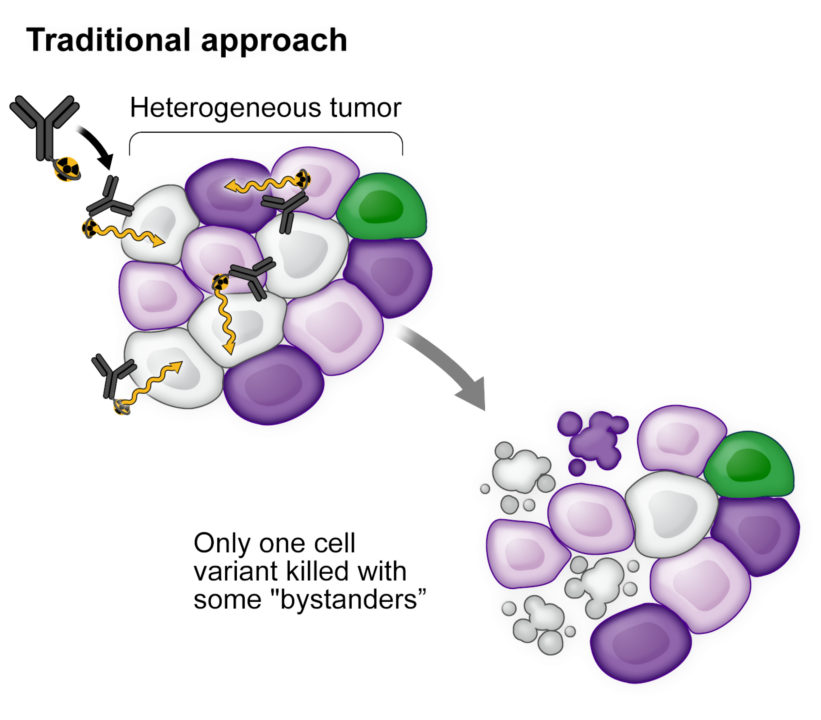
A proprietary conjugate reaches 95-97% of tumor cells. The remaining 3-5% are eradicated via the bystander effect, preventing malignant cells from mutating and regrowing.
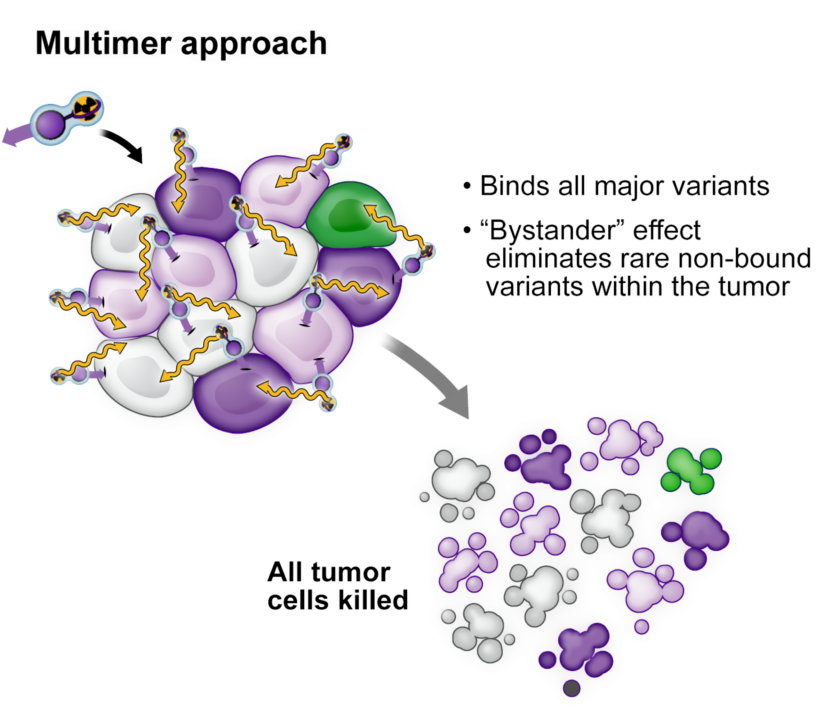
Zwitterionicity prevents off-target damage.
Excessive off-target binding and slow release of radioisotopes in the system damages healthy tissue; serious safety and tolerability issues result in abandoning therapy.
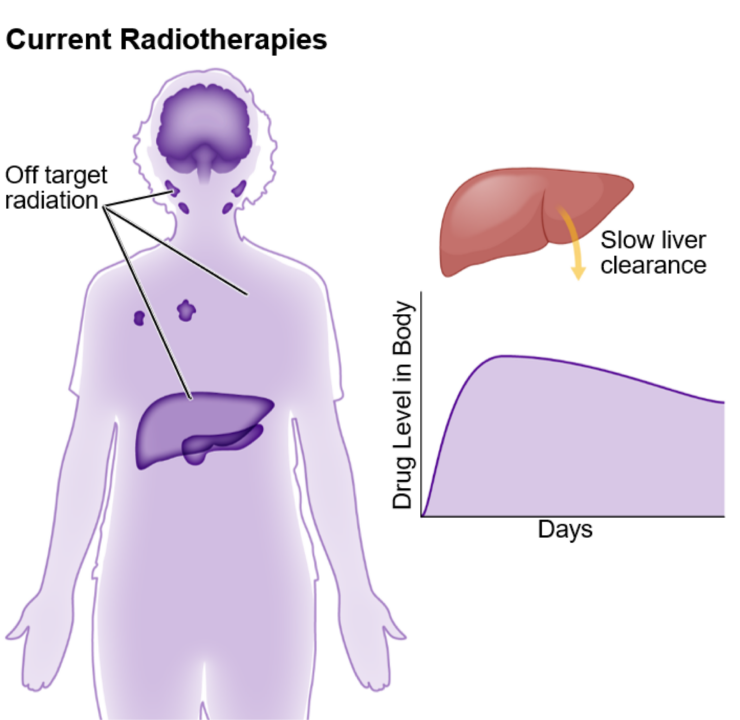
Zwitterionic core repels off-target tissues and leaves healthy cells untouched. Modeling estimates this delivers at minimum a 5-fold reduction in off-target effects as compared with current state-of the art approaches.
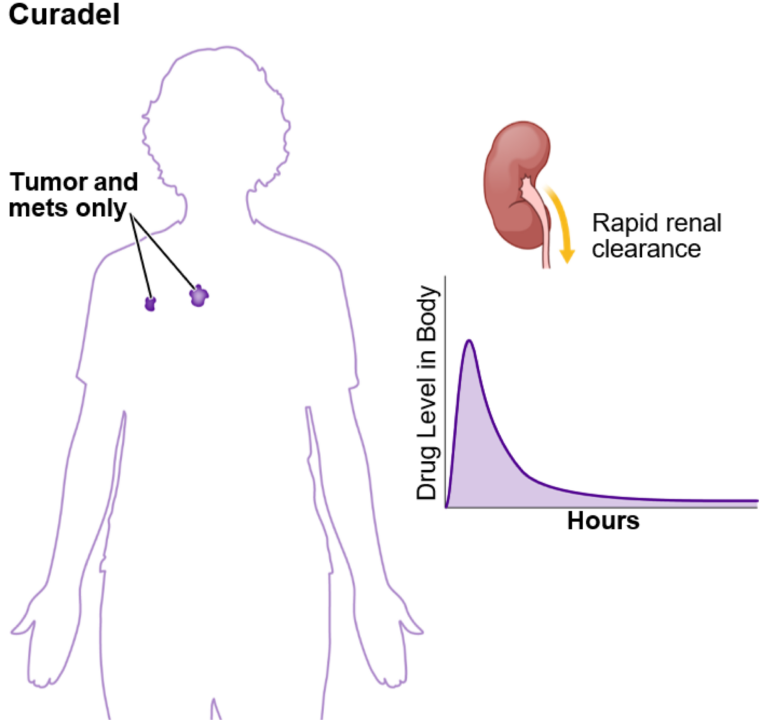
Rapid repeat dosing prevents regrowth.
Antibody drug conjugates (ADCs) and some isotopes decay and circulate for 45-60 days in the body before they can be dosed again, leaving tumors to regenerate and build resistance.
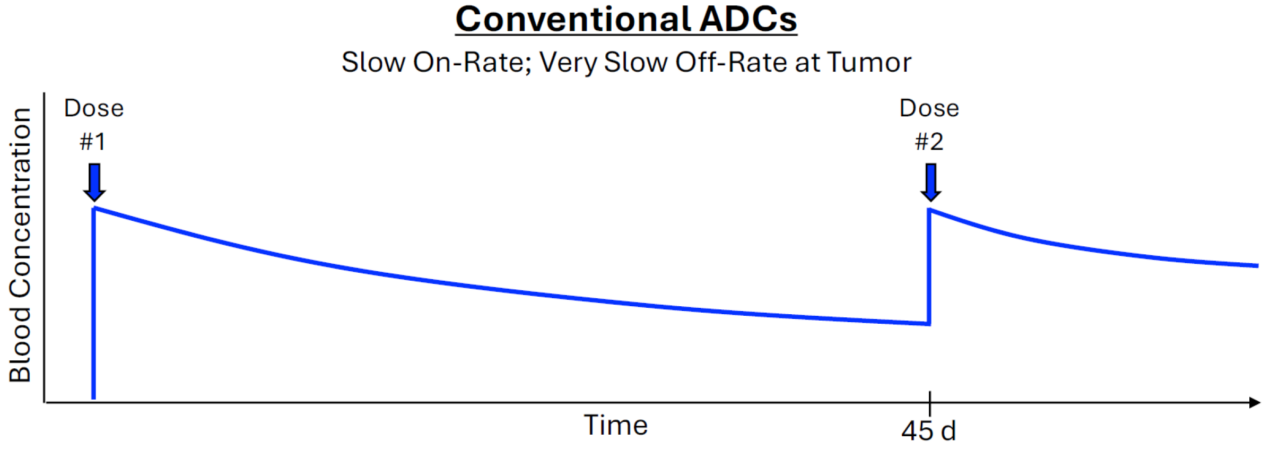
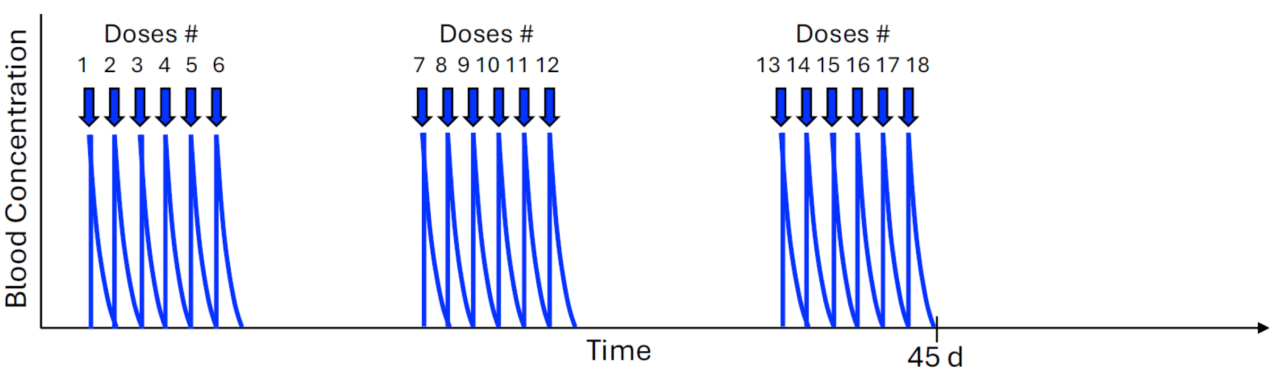
The zwitterionic drug core is cleared from the body through the kidneys in a matter of hours. This minimizes toxicity and enables redosing within one week or less to prevent tumor regrowth.
Applications for Zwitterionicity.
Our “zwitterionic hypothesis” is that designing a drug with strong polyionicity, electrically and geometrically balanced over the molecule surface, will render the drug inert in the human body while simultaneously redirecting clearance to the kidneys, where it can be eliminated rapidly. The zwitterionic hypothesis has already been proven for quantum dots (a type of nanoparticle) as well as near-infrared fluorophores (a type of small molecule). By applying the zwitterionic hypothesis to radiopharmaceuticals for targeted alpha therapy (TAT), we believe that we can revolutionize cancer treatment. In addition, Curadel has created several new families of zwitterionic metal chelators with widespread application in magnetic resonance imaging (MRI) and disease treatment. For example, CPI-005, our novel zwitterionic chelator for MRI contrast, has preliminary data indicating best-in-class relaxivity and minimal retention.